Material Compliance
Material Compliance
Would you like to find out the best way to achieve successful material compliance?
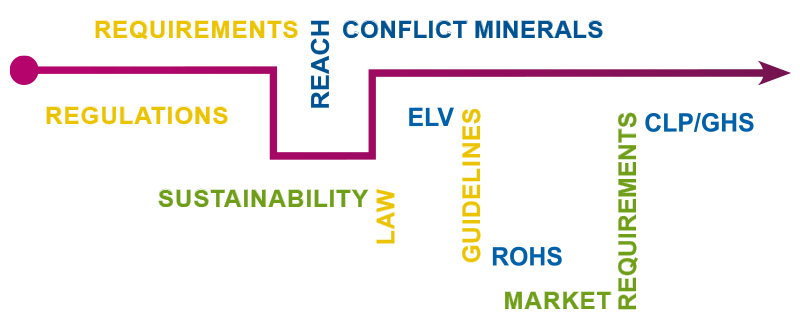
Material compliance includes adherence to material-specific requirements resulting from globally applicable laws, regulations, standards and guidelines as well as customer specifications. These include, for example, the European chemicals regulation REACH, the RoHS directive, the SCIP database, PFAS, US regulations such as Conflict Minerals, CalProp65 or the Toxic Substances Control Act (TSCA) and many more. These requirements apply to substances, mixtures and articles, regardless of whether they are available as in-house manufactured parts (products) or commercial goods.
In short: Material compliance describes the adherence to all material-specific specifications that affect your product!
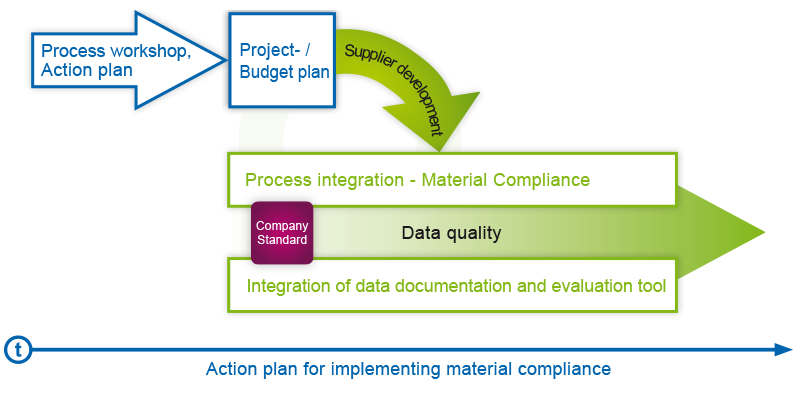
6 steps to material compliance
tec4U-Solutions has developed a 6-step plan on how manufacturers and importers of products can ensure their material compliance requirements efficiently and sustainably: